The calorific or heating value is an important indicator of the quality of the pressed fuel briquettes. It measures the energy content of the briquettes. It is defined as the amount of heat evolved when a pressed fuel briquette is completely burnt and the combustion products are cooled. And the Gross Calorific Value, shortened as GCV, refers to the calorific value with the condensation of water in the latent heat, also known as higher heating value. Whereas during combustion, the heat of condensation of water contained in the fuel and formed during combustion will become unavailable because of the vaporization of the water. And then, the useful heating value is gained after the heat of condensation of the water being subtracted from the gross calorific value, which is referred to as the Net Heating Value or lower heating value.
The calorific value of a specific pressed fuel briquette is related to the amount of oxygen required for thorough combustion. Usually, one gram of oxygen burnt, 14,022 J of energy can be released. It means the higher the oxidation of the fuel, the less the oxygen is required for thorough combustions and the lower the heating value of the fuel. Meanwhile when the fuel consisted of compounds with lower degree oxidation, such as, hydrocarbons, the heating value of the biomass will be higher.
The calorific value of the pressed fuel briquettes is limited by fuel moisture content for the reason that the heat is used to vaporized the water, lowering the heat released.
Furthermore, the calorific value of the fuel briquettes is also limited by the ash content. Approximately every 1% addition of ash translates to a 0.2 MJ/kg − 1 decrease in the heating value.
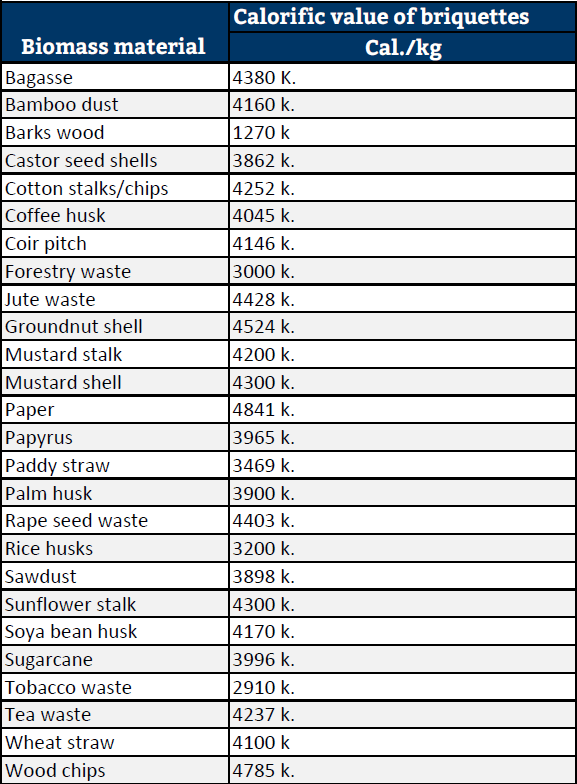
As for the calorific value of various fuel briquettes pressed from particular biomass are shown in the diagram below for your reference.

Esben M. Vestergaard
Regional Sales Manager
Do you want us to contact you?
Please fill out the form and we will get back to you.